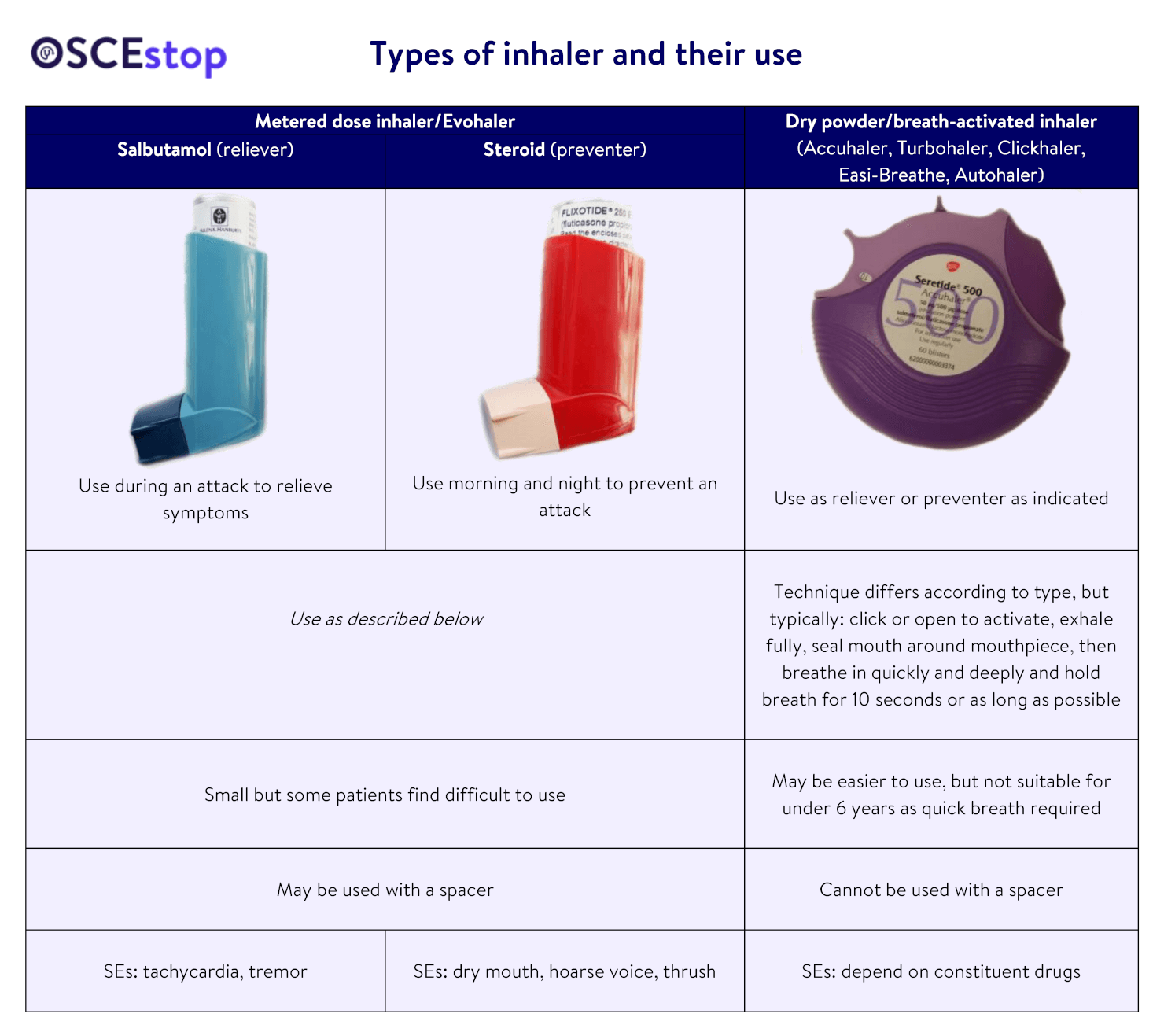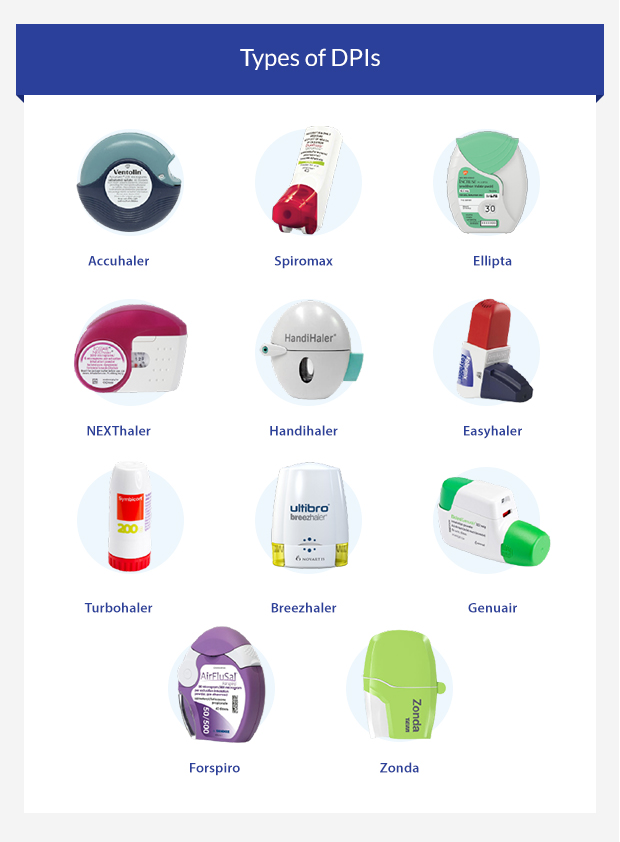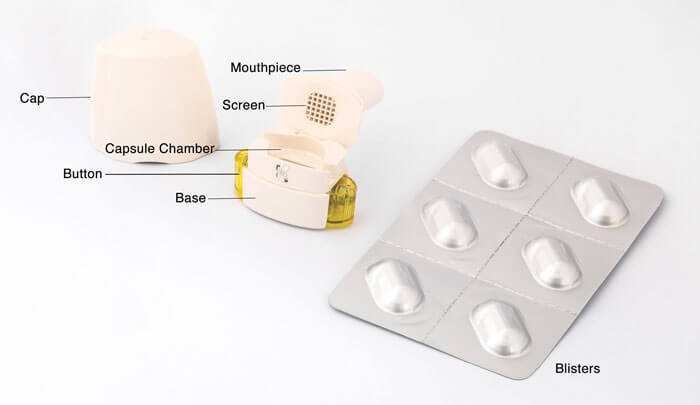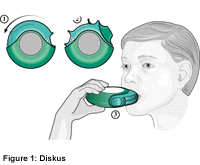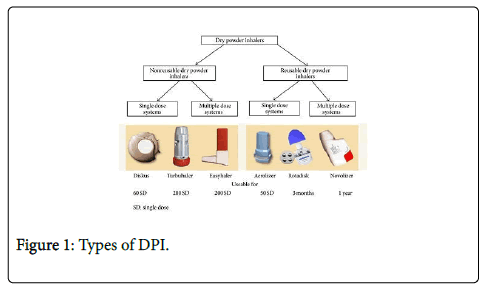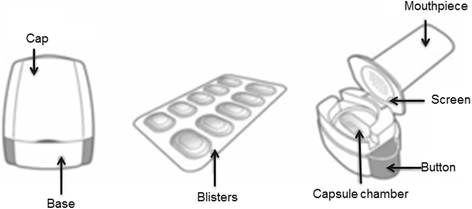
Recent advances in capsule-based dry powder inhaler technology | Multidisciplinary Respiratory Medicine | Full Text
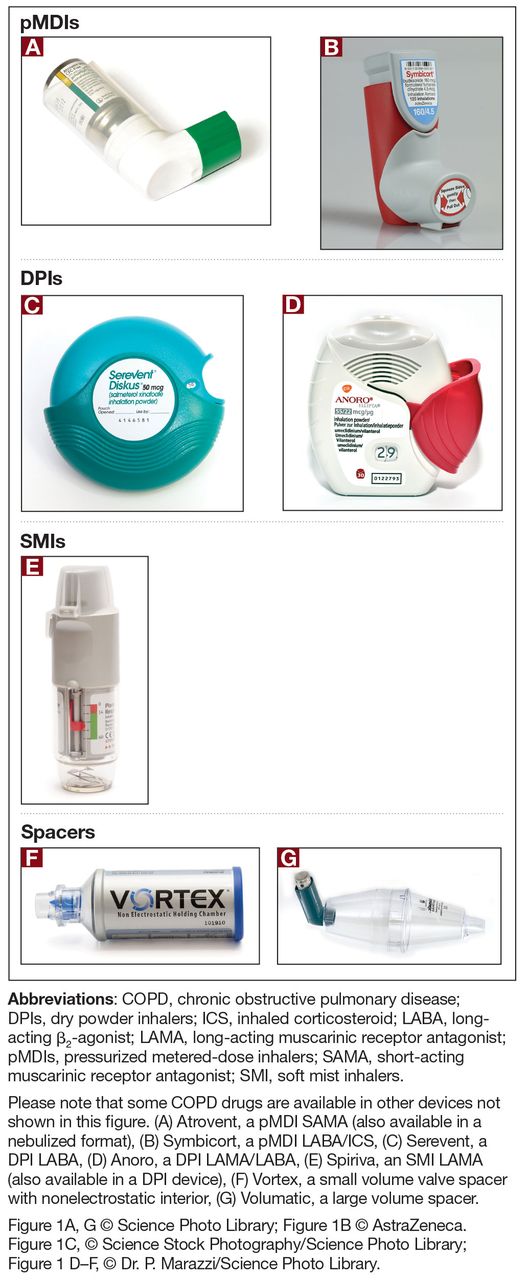
Considerations for Optimal Inhaler Device Selection in Chronic Obstructive Pulmonary Disease | Cleveland Clinic Journal of Medicine

Easyhaler Salbutamol sulfate 100micrograms/dose dry powder inhaler (Orion Pharma (UK) Ltd) 200 dose - RightBreathe
Assessment of inhalation flow patterns of soft mist inhaler co-prescribed with dry powder inhaler using inspiratory flow meter for multi inhalation devices | PLOS ONE

Scientific and regulatory activities initiated by the U.S. food and drug administration to foster approvals of generic dry powder inhalers: Quality perspective - ScienceDirect
Processes | Free Full-Text | Spray Drying for the Preparation of Nanoparticle-Based Drug Formulations as Dry Powders for Inhalation
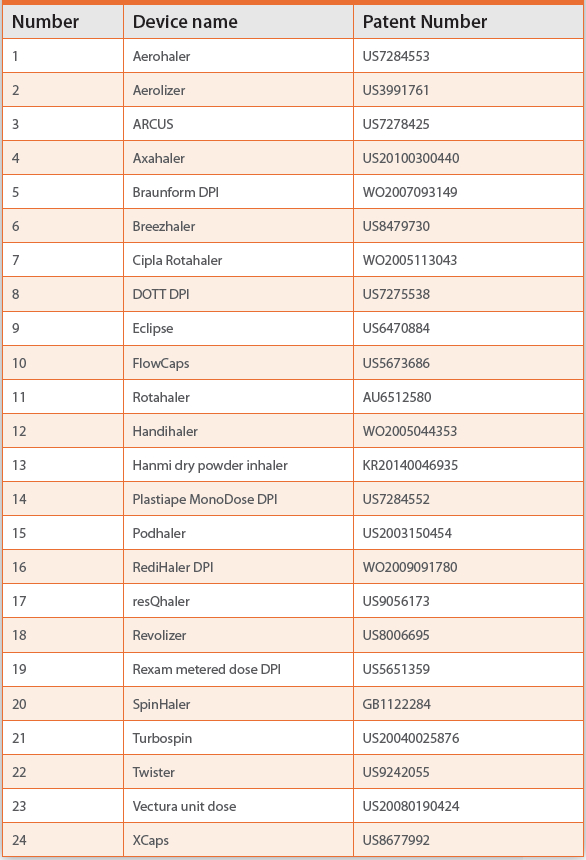
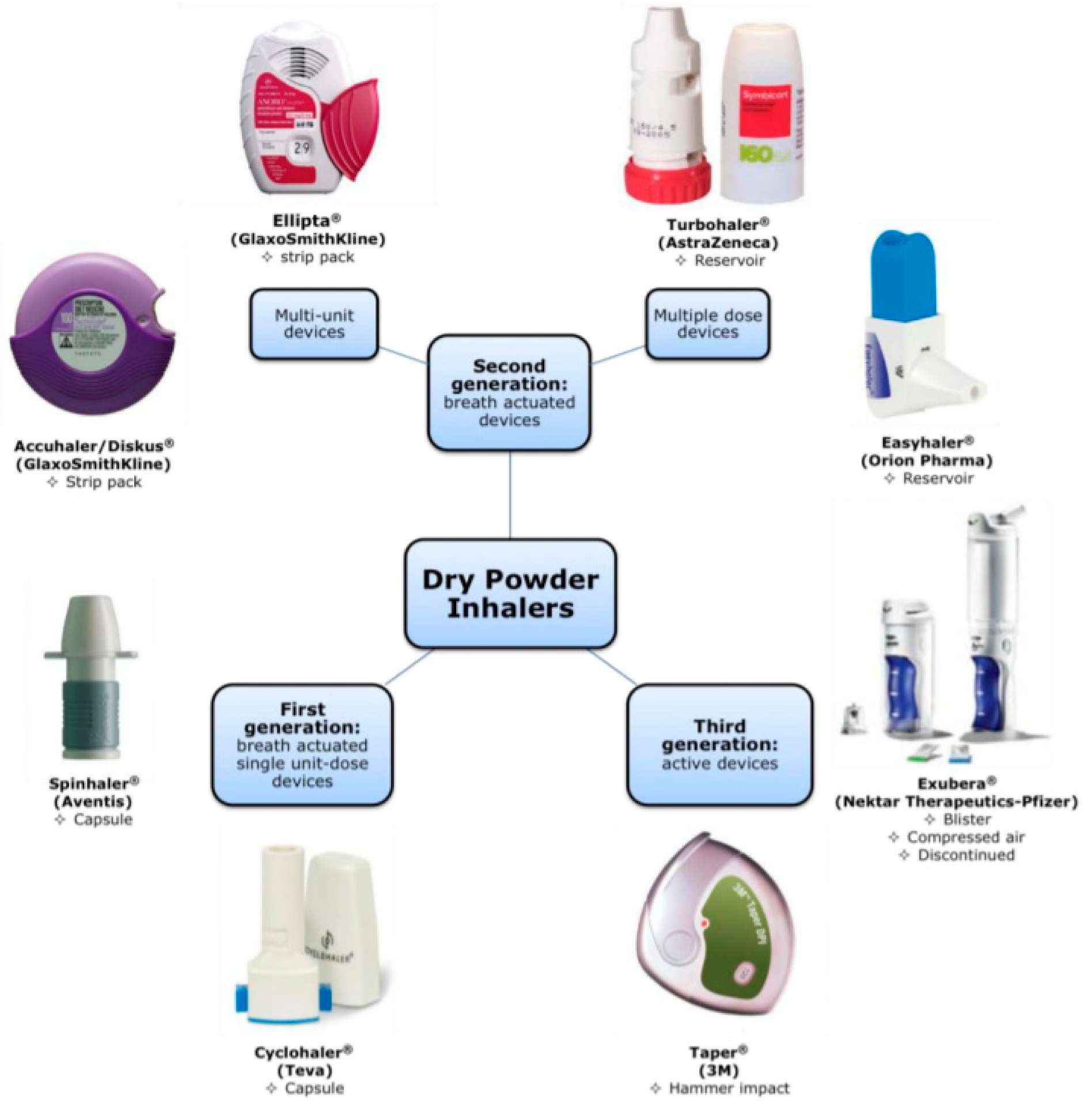
Review of Dry Powder Inhaler Devices | American Pharmaceutical Review - The Review of American Pharmaceutical Business & Technology